study material-chemistry tips tricks
Acidic Powers & Their Orders - Chemistry Fact Sheet- 1
Acidic Powers & Their Orders - Chemistry Fact Sheet- 1
| 1. | Farther the (–I) group (Cl), lesser the acidic strength | ||
| 2. | Farther the (+I) group, greater the acidic power | ||
| 3. | 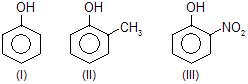 |
—CH3 is electron donating and — NO2 is electron attracting |
|
| 4. | 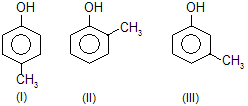 |
—CH3 is electron repelling; decreases acidic strength of phenol | |
| 5. | 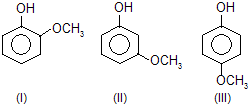 |
— OCH3 group contains +M effect and decreases acidic poer. |
|
| 6. | 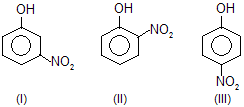 |
— NO2 is electron attracting; III is more resonance stabilised than I and also than II. In I, only inductive effect is operative. | |
| 7. | 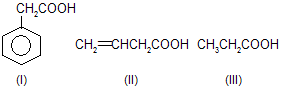 |
sp2 hybridised carbon of I, II are more el-ectronegative hence acid strength is inc-reased. Benzylic (C6H5CH2) is more stab-ilised than allylic (CH2==CHCH2). | |
| 8. | 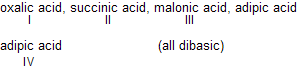 |
Effect of one —COOH on the other decr-eases as its distance between them increases, (COOH)2 is maximum acidic. | |
| 9. | —NO2 is electron attracting (–I effect) | ||
| 10. | —OH shows electron withdrawing nature at o - and m - and electron repelling at p -, o - isomer due to intramolecular bonding in salicylate ion is stronger than m - isomer | ||
| 11. | —do— | ||
| 12. | —NH2 is electron donating. |
Related Guides
- How to Prepare for IIT JEE Main 2020 during Covid
- What are the Best Books for JEE Main 2020?
- The Concepts of Chemical Equilibrium for IIT-JEE
- AIEEE 2009 Mathematics - Must have Tips and Tricks
- AIEEE 2009 Physics Last Minute Preparation Tips
- Circuit Elements, Mangetic Fields and Inductors - AC Circuits Formula Sheet for IIT-JEE and AIEEE
- Electrostatics and DC Circuits Formula Sheet for IIT-JEE and AIEEE
- Periodic Properties - QUICK notes for IITJEE and AIEEE
- Shortcuts for solving problems in AIEEE, IITJEE and other engineering exams
- Strategies of problem solving, objective type questions and others for IITJEE, MUST HAVE!!
- How to prepare for IIT JEE 2009 when you have only 4 months in your hand!
- Tips & Strategies for BITSAT, IITJEE and AIEEE 2009
- IIT JEE & AIEEE Tips for Assertion-Reason Type questions in MCQs
- Chemistry Concepts - Mole concept, Eudiometry, POAC, Limiting Reagent, Chemical Equivalence and Disproportionation reaction
- IIT-JEE Cheat Sheets - integral cheatsheet, calculus 2 cheat sheet
- The EASIEST way to crack the TOUGHEST EXAM - The IIT JEE
- IIT JEE 2010 Aspirants - Study Plan
- Top 10 Study Tips For IIT-JEE Prep
- How much YOU should study for IIT JEE
- How To Learn Organic Chemistry
- [TRICK] THE DOUBLE BOND EQUIVALENCE CONCEPT
- Step wise approach to crack IIT - JEE --- An Ex-iitians experience
- 10 golden rules to crack IIT -JEE , AIEEE and other Engineering Examinations
- Prepare for IIT - JEE : Some useful suggestions
- Start your IIT- JEE preparation today
- 10 SMALL TIPS TO CRACK AN EXAMINATION
- Expert Speak: Are you taking 11th seriously?
- CBSE and IIT-JEE are very different exams: Expert
- 7% in maths can get you through IIT-JEE
- IITJEE Simplified : Tip 3 - What is the best way to study Mathematics?
- IITJEE Simplified : Study Tip 2 - Things I should avoid?
- IITJEE Simplified: Study Tip 1 - How much should I study?
- How Can You Crack AIEEE
- Admission Help: Trends in JEE Counseling From JEE 2003 To JEE 2007
- Admission Help: What To Do After Getting Through IIT JEE
- How To Study Organic Chemistry
- Admission Help: How to choose the right Engineering Branch ?