Atomic - Structure-3
Dissimilarities between nuclear atomic model and solar system
- The sun and the planets are very bit bodies and uncharged while the nucleus and electrons are very small
- The revolution of the planets in the solar system is governed by gravitational forces, while the revolution of electrons around the nucleus is governed by electrostatic forces.
- In the solar system, there is only one planet which revolves in any particular orbit, but in the nuclear atomic model more than one electron may ratate in any particular orbit.
Drawbacks of Rutherford model
- According to classical electromagnetic theory, when a charged particle moves under the
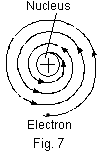 influence of attractive force, it loses energy continuously in the form of electromagnetic radiations. Thus, when the electron (a charged particle) moves in an attractive field (created by protons present in the nucleus), it must emit radiations. As a result of this, the electron should lose energy at every turn and move closer and closer to the nucleus following a spiral path (Fig. 7). The ultimate result will be that it will fall into the nucleus, thereby making the atom unstable. Bohr made calculations and pointed out that an atom would collapse in 10-8 seconds. Since the atom is quite stable, it means the electrons do not fall into the nucleus, thereby this model does not explain the stability of the atom.
influence of attractive force, it loses energy continuously in the form of electromagnetic radiations. Thus, when the electron (a charged particle) moves in an attractive field (created by protons present in the nucleus), it must emit radiations. As a result of this, the electron should lose energy at every turn and move closer and closer to the nucleus following a spiral path (Fig. 7). The ultimate result will be that it will fall into the nucleus, thereby making the atom unstable. Bohr made calculations and pointed out that an atom would collapse in 10-8 seconds. Since the atom is quite stable, it means the electrons do not fall into the nucleus, thereby this model does not explain the stability of the atom. - If the electrons lose energy continuously, the observed spectrum should be continuous but the actual observed spectrum consists of well defined lines of definite frequencies. Hence, the loss of energy by the electrons is not continuous in an atom.
3. Atomic terms
- Atomic Number (Z)
The atomic number of an element is the number of protons contained in the nucleus of the atom of that element. - Nucleons
Protons and neutrons are present in a nucleus, so these fundamental particles are collectively known as nucleons. - Mass Number (A)
The total number of protons and neutrons i.e., the number of nucleons present in the nucleus is called the mass number of the element. - Nuclide
Various species of atoms in general. A nuclide has specific value of atomic number and mass number.
IUPAC notation of an atom ( nuclide)
Let X be the symbol of the element, its atomic number be Z and mass number be A. Then the element can be represented as zXA. - lsotopes
Atoms of the element with same atomic number but different mass number e.g.1H1,1H2. There are three isotopes of hydrogen. - Isobars
Atoms having the same mass number but different atomic numbers, e.g.15P32 and 16S32 called isobars. - isotones
Atoms having the same number of neutrons but different number of protons or mass number, e.g.6C14,8O16 ,7N15 are called isotones. - isoelectronic
Atoms, molecules or ions having same number of electrons are isoelectronic e.g. (a) N2CO,CN-(b) N-3, O-2, F-. - Nuclear isomers (isomeric nuclei) are the atoms with the same atomic number and same mass number but with different radioactive properties.
Example of nuclear isomers isCH ºº CH and N2 are isoelectronic because they contain same number of electrons (14) but they are not isosters whole benzene (C6H6) and Inorganic benzene (B3N3H6) borazine are isosters because both contain same number of electrons and atoms.
Uranium-X( half life 1.4 min) and
Uranium-Z ( half life 6.7 hours)
The reason for nuclear isomerism is the different energy states of the two isomeric nuclei.
Other examples are
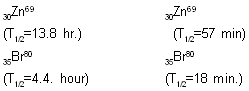
- Isosters
Molecules having same number of atoms and also same number of electrons are called isosters.
E.g.,- N2 and CO
- CO2 and N2 O
- HCI and F2
- Atomic mass unit
It is exactly equal to 1/12 of the mass of 6C12atom.
1amu = 1.66 x 10-27 Kg. = 931.5 MeV
Nature of Electromagnetic Radiation (Electromagnetic wave Theory)
This theory was put forward by James Clark Maxwell in 1864. The main points of this theory are summed up as follows :
- The energy is emitted from any source (like the heated rod or the filament of a bulb through which electric current is passed) continuously in the form of radiations (or waves) and is called the radiant energy.
- The radiations consist of electric and magnetic fields oscillating perpendicular to each other and both perpendicular to the direction of propagation of the radiation.
- The radiations possess wave character and travel with the velocity of light (i.e. nearly 3 × 108 m/sec).
Because of the above characteristics, the radiations are called Electromagnetic radiations or Electromagnetic waves. - These waves do not require any material medium for propagation. For example, rays from the sun reach us through space which is a non-material medium.
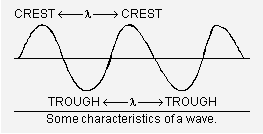
Some important characteristics of a wave. The main characteristics of a wave are its wavelength (l), frequency (n) and velocity (c). These are defined as follows :
1 Å = 10-8 cm = 10-10 m
1 nm = 10-9 m, 1 pm = 10-12 m
1 Hz = 1 cycle/sec
Besides the above three characteristics, two other characteristics of a wave are amplitude and wave number.
![]()
| Among various forms of visible light, violet colour has shortest wave length, highest freqiency and highest energy while red coloured light has borqest wavelength and least frequency. |
If ![]() is expressed in cm,
is expressed in cm, ![]() will have the units cm-1
will have the units cm-1
Relationship between velocity, wavelength and frequency of a wave. As frequency is the number of waves passing through a point per second and ![]() is the length of each wave, hence their product will give the velocity of the wave. Thus
is the length of each wave, hence their product will give the velocity of the wave. Thus
c = n ×![]()
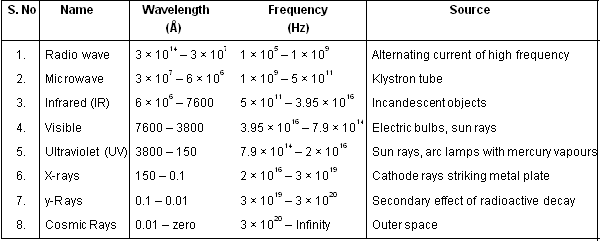
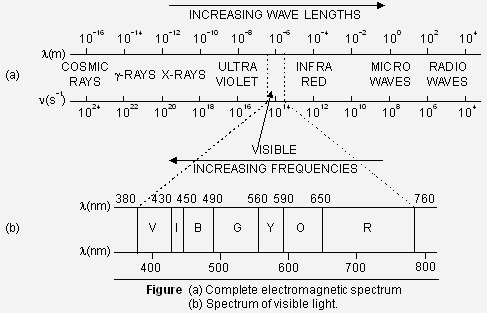
Electromagnetic spectrum. The different types of electromagnetic radiations differ only in their wavelength and hence frequencies. Their wavelengths increase in the following order :_
Cosmic rays < ![]() -rays < X-rays < Ultra-violet rays < Visible < Infrared < Micro waves < Radio waves
-rays < X-rays < Ultra-violet rays < Visible < Infrared < Micro waves < Radio waves
Limitations of Electromagnetic Wave Theory. Electromagnetic wave theory was successful in explaining the properties of light such as interference, diffraction etc. but it could not explain the phenomena of `Black body radiation', and `Photoelectric effect' the discussion of which is beyond the scope of the book.
Study of Emission And Absorption Spectra
We have studied above that the electromagnetic spectrum consists of radiations of different wave lengths and frequencies. An instrument used to separate the radiations of different wavelengths (or frequencies) is called spectroscope or a spectrograph. A spectroscope consists of a prism or a diffraction grating for the dispersion of radiations and a telescope to examine the emergent radiations with the human eye. However, if in a spectroscope, the telescope is replaced by a photographic film, the instrument is called a spectrograph and the photograph (or the pattern) of the emergent radiation recorded on the film is called a spectrogram or simply a spectrum of the given radiation.
Depending upon the source of radiation, the spectra are broadly classified into (i) Emission spectra and (ii) Absorption spectra. These are briefly explained below :–
1. Emission spectra. When the radiation emitted from some source e.g. from the sun or by passing electric discharge through a gas at low pressure or by heating some substance to high temperature etc. is passed directly through the prism and then received on the photographic plate, the spectrum obtained is called ‘Emission spectrum’.
The emission spectra are mainly of two types:
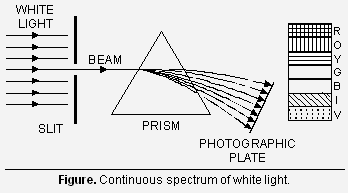
(i) Continuous spectra When white light from any source such as sun, a bulb or any hot glowing body is analysed by passing through a prism, it is observed that it splits up into seven different wide bands of colours from violet to red, (like rainbow), as shown in Figure. These colours are so continuous that each of them merges into the next. Hence the spectrum is called continuous spectrum.
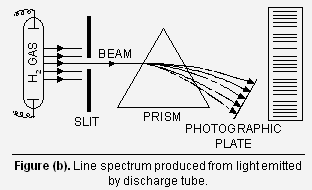
(ii) Line spectra When some volatile salt (e.g., sodium chloride) is placed in the Bunsen flame (Fig. a) or an electric discharge is passed through a gas at low pressure (Fig. b), light is emitted. If this light is resolved in a spectroscope, it is found that no continuous spectrum is obtained but some isolated coloured lines are obtained on the photographic plate separated from each other by dark spaces. This spectrum is called ‘Line spectrum’.
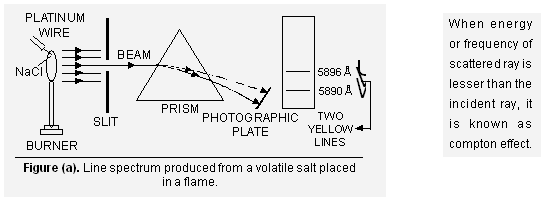
Each line in the spectrum corresponds to a particular wavelength. Further it is observed that each element gives its own characteristic spectrum, differing from those of all other elements. For example, sodium always gives two yellow lines (corresponding to wavelengths 5890 and 5896 Å). Hence the spectra of the elements are described as their finger prints differing from each other like the finger prints of the human beings.
Further, it will be discussed later that the line spectra are obtained as a result of absorption and subsequent emission of energy by the electrons in the individual atoms of the element. Hence the line spectrum is also called atomic spectrum.
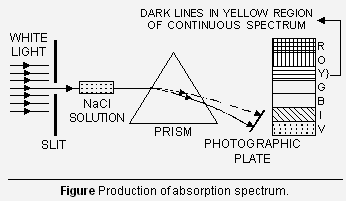 2. Absorption spectra When white light from any source is first passed through the solution or vapours of a chemical substance and then analysed by the spectroscope, it is observed that some dark lines are obtained in the otherwise continuous spectrum (Figure). These dark lines are supposed to result from the fact that when white light (containing radiations of many wavelengths) is passed through the chemical substance, radiations of certain wavelengths are absorbed, depending upon the nature of the element. Further it is observed that the dark lines are at the same place where coloured lines are
2. Absorption spectra When white light from any source is first passed through the solution or vapours of a chemical substance and then analysed by the spectroscope, it is observed that some dark lines are obtained in the otherwise continuous spectrum (Figure). These dark lines are supposed to result from the fact that when white light (containing radiations of many wavelengths) is passed through the chemical substance, radiations of certain wavelengths are absorbed, depending upon the nature of the element. Further it is observed that the dark lines are at the same place where coloured lines are
obtained in the emission spectra for the same substance. This shows that the wavelengths absorbed were same as were emitted in the emission spectra. The spectrum thus obtained is, therefore, called ‘absorption spectrum’.
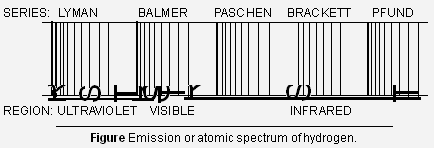
Emission spectrum of Hydrogen When hydrogen gas at low pressure is taken in the discharge tube and the light emitted on passing electric discharge is examined with a spectroscope, the spectrum of hydrogen (Fig. b). It is found to consist of a large number of lines which are grouped into different series, named after the discoverers. The names of these series and the regions in which they are found to lie are given in the Figure below.
In case of light, some phenomena like interference, diffraction etc. can be explained if the light is supposed to have wave character. However, certain other phenomena such as black body radiation and photoelectric effect can be explained only if it is believed to be a stream of photons i.e., has particle character (or is corpuscular in nature). Thus, light is said to have a dual character. Such studies on light were made by Einsterin in 1905.
Louis de Broglie, a French physicist, in 1924, advanced the idea that like photons, all material particles such as electron, proton, atom, molecule, a piece of chalk, a piece of stone or an iron ball (i.e. microscopic as well as macroscopic objects) possessed both wave character as well as particle character. The wave associated with a particle is called a matter wave.