The Concepts of Chemical Equilibrium for IIT-JEE
- Illustrartion
- 0.0753 g of selenium vapours occupiedmol of 78ml at STP. Selenium is in state of equation.
Se6(g) 3Se2(g)
3Se2(g)
Calculation deg. of dissociation
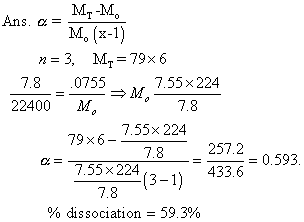
Factors affecting equation:-
(1) Chyange of conc. Of any reactant or product
(2) Change in temp. of system
(3) Change in pressure of system
(4) Tradition of catalyst
(5) Addition of inert gas.
Effect of addition of catalyst
:- Addition of catalyst does not disturb eqn. But helps in attainment of eqn. quickly.
(a)
If rxn takes place at constant vol.(closed vessel)
:- Addition of inert gas will not chang mole conc. Of reactants & products. So, state of eqn. Will remain unaffected.
(b) If rxn. Takes place at constant pressure, Hence dissociation of pcl3 increases.
Dumb quation:-[Q. How this happen?
Ans. Since p is cnstant, so addition of inert gas will Total volume. So, at equation, molec conc of each reactant old products will . So,kc is constant at constant temp.
So, to keep kc constant either [PCl5] should or [PCl3] & [Cl2] should
should or [PCl3] & [Cl2] should . This happen only if PCl5dissociates.
. This happen only if PCl5dissociates.
Le chathelier’s principle:-
If a in eqn. Is subjected to a change of conc. Temp. of pressure, eqn. Shifts in direction that tends to undo the of the change imposes.
(1) Effect of conc.:-
A + B C + D
C + D
If rxn is in eqn. Mole of A or B is added, then ,according to Le chatelic’s principle effect will be to
conc. Of A & B react to frome C & D. I,e;eq m. shifts in forward direction. Shift in forward.
(2) Effect of temp.:-
Every rxn. is made up of two opposing rxns. If to rewards rxn. Is exothermic, then backwardrxn.
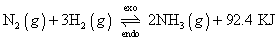
If temp. is
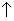
,I,e; heat is supplied to system; according to the chateliec princple. i,e; eqn. Will shift to side heat absorbs heat I,e; in backward direction & vice versa.
Note
:- Exothermic rxn are forward by low temp. whereas end. thermic rxn. are favoured by high temp.
(3) Effect of pressure:-
Significant role to play only in case of gaseons rxn.
N2O4(g) 2NO2(g)
2NO2(g)
1 mole 2 mole
Forward rxn. Occurs with
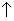
in no. of moles & viseversa. If pressure on system
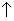
, then according to Le chateliec’s principle , eqn. Will shift in direction in which pressure
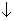
or no of moles (b/c pressure is directly proportional to no. of moles). Since backward rxn. Takes place with in no of. moles. So,
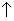
in pressure will favovr. Combination of NO2molecules to N2O4.
Note
:-
(1) Low pressure tavours those rxns which accompany by
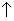
in total no of moiles & vice versa.
(2) Pressure has no effect on eqn. Rxn which proceed which no change in total no. of moles. So,
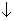
in volume will shift eqn. In direction in which no of moles decreases.
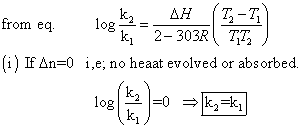
I,e; eqn. Constant does not change with temp.
(ii)
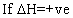 log(k2/k1)= +velog k2>log k1
log(k2/k1)= +velog k2>log k1 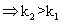 I,e; eqn. Constant
I,e; eqn. Constant with
with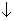 in temp.
in temp.
(iii)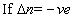 log(k2/k1) = -velog k2< log K1
log(k2/k1) = -velog k2< log K1  I,e; eqn. Constant
I,e; eqn. Constant with
with in temp.
in temp.
Q.1 Calculate eqn. constant for rxn.
H2(g) + CO2(g) H2
H2
O(g) + CO(g) at 1539K, if eqn. constants at 1395K for following are
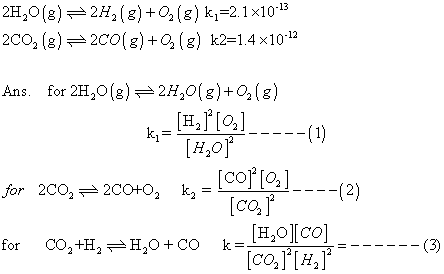
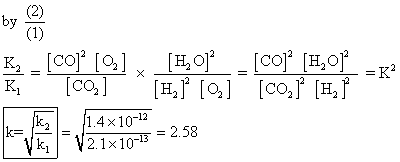
Q2.for reaction
A + B 3C at 290
3C at 290
C, 3L vessel contains 1,2,4 mole A, B & c resp. predict direction of rxn .
if (a)kcfor rxn. is 10
(b) kcfor rxn is 10.66
Ans.
:- A + B 3C
3C
Before rxn; [A] = 1⁄3
[B] = 2⁄3
[C]= 4⁄3
so,
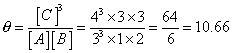
and this [c] must
or [A] & [B] should so, rxn mole occurs in backward direction
(b) Q = Kc
, So, rxn. is in eqn.
Q.3 At temp, 2AB2(g) 2AB(g) + B2(g)
2AB(g) + B2(g)
with deg association ‘x’ which is small compared to unity. Device expression for ‘x’ in terms of eqns. Const. Kp& total pressure P.
Ans. 2AB2(g) 2AB(g) + B2(g)
2AB(g) + B2(g)
Mole after dissociation

Mole after dissociation
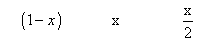
Total mole at eqn.
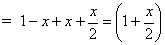
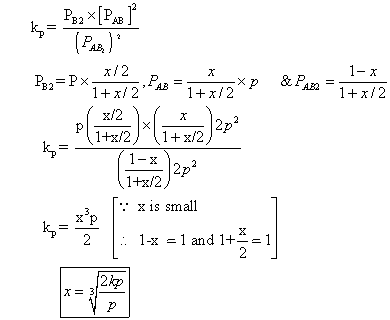
Q.4:- kCFOR a2(G) + b2(g) 2AB (g) at 1000
2AB (g) at 1000
C is 50. If one line flask containing 1 mole of A2 is connected with a 2L flask containing
2mole of B2 ; How many mole of AB will be formed at
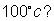
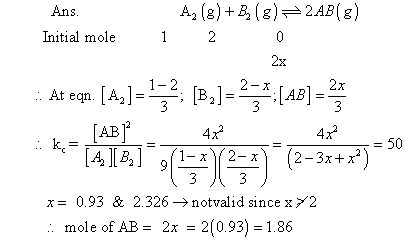
Q.5 N2(g) + O2(g) 2NO(g)
2NO(g)
was established with an eqn. constain K
c= 2.1 X 10- 3. At eqn. mole % of No. was 1.8. estimates initial composition of air in mole fraction of N2& O2.
Ans: N
2
(g) + O2 2NO
2NO
initial a (100 - a) 0
final (a - x) (100 - a - x) 2n
given
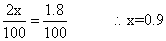
also
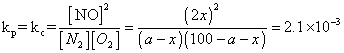
a = 79%, 100 - a = 21%
Q.6: kpfor rxn. N2+ 3H2 2NH3is 1.6 X 10-4atm-2at 4000c what will be kpat 5000
2NH3is 1.6 X 10-4atm-2at 4000c what will be kpat 5000
C ? Heat of rxn. in this temp. range is - 25.14 KCal.
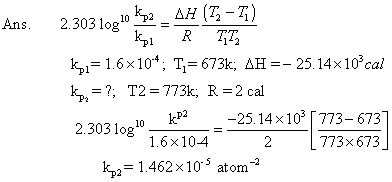
Out Kpfor rxn. Also report Kp& G0forN2+ 3H2
G0forN2+ 3H2  2NH3at 250C
2NH3at 250C
Ans. -
 G0= 2.303 RT log10Kp- ( - 16.5 X 103) = 2.303 X 8.314 X 298 log10Kp
G0= 2.303 RT log10Kp- ( - 16.5 X 103) = 2.303 X 8.314 X 298 log10Kp
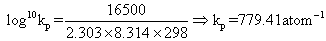
Kp1for N2+ 3H2 2NH3Kp1= (Kp)2= (779.41)2= 6.07 X 105atm- 1Also
2NH3Kp1= (Kp)2= (779.41)2= 6.07 X 105atm- 1Also
 G01= 2.303 X 8.314 X 298 log10(6.07 X 105) J
G01= 2.303 X 8.314 X 298 log10(6.07 X 105) J
=32.998 KJ
 G01= -32.998 KJ
G01= -32.998 KJ
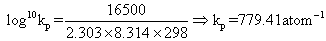
Q.9 At S4 Ok & 1 atom, N2O4is 66% dissociated into NO2
what mol. of 10g N2O4occupy under these condition
Ans:- N2O4 2NO2
2NO2
initial 1 0
at eqn
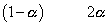
total mole at eqn
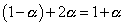
but
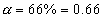
Total mole of eqn. = 1 + 0.66 = 1.66
1 mole of N2O4
is taken, mole at eqn.=1.66
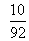
mole of n2o4 is taken ,mole at eqn. =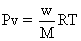
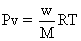
1 X v = 0.18X0.0821X340 = 5.02 L
Q.10 heat of rxn for rxn. At constant vol. is 1200 cal more than at constant pressure at 300k. calculate ratio of eqn. Constant kp& kc.
Ans.
 E =
E = H = 1200 Cal. Whrere
H = 1200 Cal. Whrere
 E is heat of rxn. at constant volume &
E is heat of rxn. at constant volume &
 H is heat of rxn. at constant pressure
H is heat of rxn. at constant pressure
 H =
H = E +
E + nRT
nRT
 nRT = -1200
nRT = -1200
 n = -1200 / 2X300 = -2
n = -1200 / 2X300 = -2
Kp= Kc(RT) n
n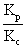 = (0.0821 X 300)-2= 1.648 X 10-3
= (0.0821 X 300)-2= 1.648 X 10-3
Q.1. 0.96g of HI sere heated to attom eqn. 2HI H2+ I2
H2+ I2
rxn. mixture on titration required 15.7 ml of N/10 hypo sol. Calculate deg. of dissociation of HI.
Ans. 2HI H2+ I2
H2+ I2
mole at t = 0
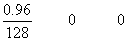
= 7.5 X 10
-3
at eqn.
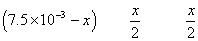
meg. Of I2
formed at eqn. = meg.of hypo used
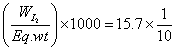
 Eqn. of I2
Eqn. of I2
=
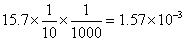
Mole of I
2
formed =
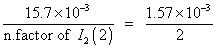
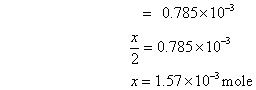
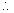 Deg. of dissociation of HI =
Deg. of dissociation of HI =
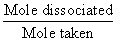
=
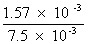
= 0.209 = 20.9 %
Q.2 An eqn. Mixture of
CO(g) + H2O(g) CO2(g) + H2(g)
CO2(g) + H2(g)
present in a vessel of 1 L capacity at 815c was found by analysis to contain 0.4 mole of Co, 0.3 mole of H2O, 0.2 mole of Co2and 0.6 mole of H2
(a) Calculate kc
(b) To conc. to 0.6 mole by adding co2to vessel, how many mole must be added into eqn mix at mole at eqn 0.4 0.3 0.2 0.6
conc. to 0.6 mole by adding co2to vessel, how many mole must be added into eqn mix at mole at eqn 0.4 0.3 0.2 0.6
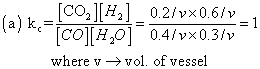
(b) Suppose ‘a’ mole of CO2 are forced in vessel at eqn.; by doing so rxn proceeds in backward direction
i.e Co2+ H2 CO + H2O
CO + H2O
at intial time (0.2 + a ) 0.6 0.4 0.3
at eqn. (0.2+a-0.2) (0.6-0.2) (0.4+0.2) (0.3+0.2)
a 0.4 0.6 0.5
Q.3 Deg of dissociation is 0.4 at 400k and 1 atom for gaseous rxt PCl
s PCL3+ CL2
PCL3+ CL2
Assume ideal gas calculate density of eqn mixture at 400k and 1 atm.
(Atomic mass of p=31& cl=35.5)
Ans: PCl
s PCL3+ CL2
PCL3+ CL2
intial mole 1 0 0
At eqn. (1-0.4) 0.4 0.4
Total mole at eqn. = 1 - 0.4 + 0.4 + 0.4 = 1.4
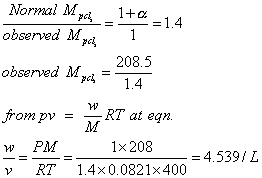
Q.5 Heat enthalpy standard free energy change for
eqn. Zn(S) + H2O(g) ZnO(s) + H2(g)
ZnO(s) + H2(g)
eqn Are 224 kJ mol
-1
and 33kjmol respectively at temp 920 calculate temp at which eqn constant becomes greater than
(1) Assume
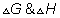
remains constant.
Ans.
 G0= -2.303 RT logK
G0= -2.303 RT logK
33000 = -2.303 X 8.314 X 920 log k
k920k= 1.34 x 10- 2
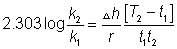
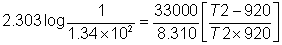
T2= 1078.5 K.
SO, At any temp above this k will be greater than
Hard type
Q.1:- Solid nh4 on rapid healthy in aclosed vessel at 357 develop a constant pressure of 275 mmHg owing to partial decomposition of NH4I into NH
3& HI . calculate final pressure developed at eqn.kpfor dissociation of HI is 0.015 at 357 C
Ans:- NH4I(g) NH3(g) + HI(g)—–(1)
NH3(g) + HI(g)—–(1)
for this eqn 2p=275 –> p=137.5
So kpfor eq. (1)
Kp= PNH3XPHI= 137.5 X 137.5 ———(2)
For 2HI H2+ I2
H2+ I2
1 0 0
(1 - x) x/2 x/2
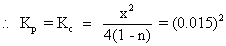
Dumb question - why we have taken kp (0.015)2?
Ans: K
pfor division of HI = 0.015
HI

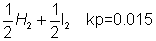
K
p
= 0.015
2HI H2+ I2 Kp= (0.015)2x = 0.2
H2+ I2 Kp= (0.015)2x = 0.2
Again since HI decomposes rxn proceeds in forward directioal (by Leehatetics Principal)
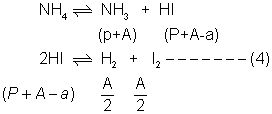
But a = (P + A) X 0.2
also kp
for final eqn (3)
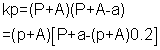
= 0.8(P + A)2But Kp
= 137.5 X 137.5
0.8(P + A)2
= 137.5 X 137.5
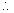 P + A = 153.73
P + A = 153.73
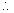 A = 16.22
A = 16.22
a = (p + A) X 0.2 = 153.73 X 0.2 = 30.75
Total pressure at eqn =PNH3+PHI+PH2+PI
=(P+A)+(P+A-a)+A/2+A/2
=2p+2A
=2(p+A)= 2x153.73
=307.46 mmHg
Q.2: when 1 - Pentyne (a)is treated with 4n atc. KOH at 175 it slowely get converted into an eqn mix of 1.3 % 1 pentyne 95.2% 2 pentyne(b) of 1,2 pentadiene ©eqn. was maintained at 175c calculate
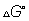
for following eqn.
from calculated value of g1 and g2 indicate order of (a) (b)© write a reasonable rxn mechanism in showing all intermediates reading to (A)(B)©

= 16.178 KJ
Stability order for A & B is B > A
Similerly for B
 C
C
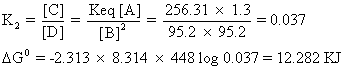
So, stability order for B and Cis B > C
Total stability order is B><>A
Q3. N2& 02
combine at a giventemp to produce no. at eqn the yield of no is x% by volume. If x =
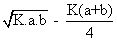
where k is is eqn constant given rxn. at given temp and a and b volume % of n2 &o2 respectively in intial pure mixture what should be the intial composition of reacting mixture in order that max. yield of nois ensured? also repo max value of k at which x is maximum?
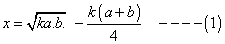
‘x’ is maximum only when condition of maximum are fulful
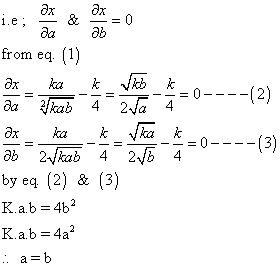
Note: this valid only when k<4 because if a = b Eq.(1)
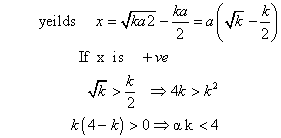
Equilibrium
Physical Eqn.
Low of mass action
Law of chemical Eqn.
Eqn.constant
Revessible rxn.
Forward rxn
Backward rxn
Gibbs free energy and Spontaneity
Homogeneous Equilibrium
Heterogeneous Equilibrium Le chatelier’s principleSource: goiit.com
+ CO(g) at 1539K, if eqn. constants at 1395K for following are<br />
- Dumb question
- why we have taken kp (0.015)2?
Ans: K
pfor division of HI = 0.015
HI

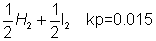
K
p
= 0.015
2HI H2+ I2 Kp= (0.015)2x = 0.2
H2+ I2 Kp= (0.015)2x = 0.2
Again since HI decomposes rxn proceeds in forward directioal (by Leehatetics Principal)
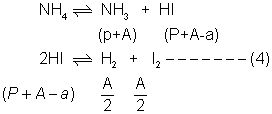
But a = (P + A) X 0.2
also kp
for final eqn (3)
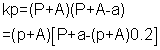
= 0.8(P + A)2But Kp
= 137.5 X 137.5
0.8(P + A)2
= 137.5 X 137.5
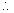 P + A = 153.73
P + A = 153.73
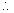 A = 16.22
A = 16.22
a = (p + A) X 0.2 = 153.73 X 0.2 = 30.75
Total pressure at eqn =PNH3+PHI+PH2+PI
=(P+A)+(P+A-a)+A/2+A/2
=2p+2A
=2(p+A)= 2x153.73
=307.46 mmHg
Q.2: when 1 - Pentyne (a)is treated with 4n atc. KOH at 175 it slowely get converted into an eqn mix of 1.3 % 1 pentyne 95.2% 2 pentyne(b) of 1,2 pentadiene ©eqn. was maintained at 175c calculate
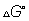
for following eqn.
from calculated value of g1 and g2 indicate order of (a) (b)© write a reasonable rxn mechanism in showing all intermediates reading to (A)(B)©

= 16.178 KJ
Stability order for A & B is B > A
Similerly for B
 C
C
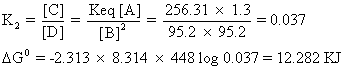
So, stability order for B and Cis B > C
Total stability order is B><>A
Q3. N2& 02
combine at a giventemp to produce no. at eqn the yield of no is x% by volume. If x =
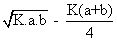
where k is is eqn constant given rxn. at given temp and a and b volume % of n2 &o2 respectively in intial pure mixture what should be the intial composition of reacting mixture in order that max. yield of nois ensured? also repo max value of k at which x is maximum?
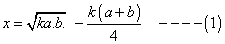
‘x’ is maximum only when condition of maximum are fulful
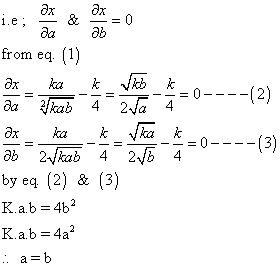
Note: this valid only when k<4 because if a = b Eq.(1)
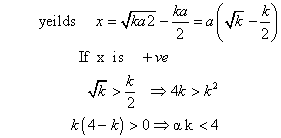
Equilibrium
Physical Eqn.
Low of mass action
Law of chemical Eqn.
Eqn.constant
Revessible rxn.
Forward rxn
Backward rxn
Gibbs free energy and Spontaneity
Homogeneous Equilibrium
Heterogeneous Equilibrium Le chatelier’s principleSource: goiit.com
temp. range is - 25.14 KCal.<br />
- Dumb question
- why we have taken kp (0.015)2?
Ans: K
pfor division of HI = 0.015
HI

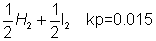
K
p
= 0.015
2HI H2+ I2 Kp= (0.015)2x = 0.2
H2+ I2 Kp= (0.015)2x = 0.2
Again since HI decomposes rxn proceeds in forward directioal (by Leehatetics Principal)
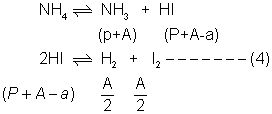
But a = (P + A) X 0.2
also kp
for final eqn (3)
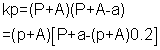
= 0.8(P + A)2But Kp
= 137.5 X 137.5
0.8(P + A)2
= 137.5 X 137.5
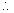 P + A = 153.73
P + A = 153.73
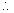 A = 16.22
A = 16.22
a = (p + A) X 0.2 = 153.73 X 0.2 = 30.75
Total pressure at eqn =PNH3+PHI+PH2+PI
=(P+A)+(P+A-a)+A/2+A/2
=2p+2A
=2(p+A)= 2x153.73
=307.46 mmHg
Q.2: when 1 - Pentyne (a)is treated with 4n atc. KOH at 175 it slowely get converted into an eqn mix of 1.3 % 1 pentyne 95.2% 2 pentyne(b) of 1,2 pentadiene ©eqn. was maintained at 175c calculate
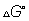
for following eqn.
from calculated value of g1 and g2 indicate order of (a) (b)© write a reasonable rxn mechanism in showing all intermediates reading to (A)(B)©

= 16.178 KJ
Stability order for A & B is B > A
Similerly for B
 C
C
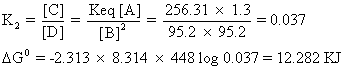
So, stability order for B and Cis B > C
Total stability order is B><>A
Q3. N2& 02
combine at a giventemp to produce no. at eqn the yield of no is x% by volume. If x =
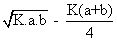
where k is is eqn constant given rxn. at given temp and a and b volume % of n2 &o2 respectively in intial pure mixture what should be the intial composition of reacting mixture in order that max. yield of nois ensured? also repo max value of k at which x is maximum?
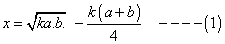
‘x’ is maximum only when condition of maximum are fulful
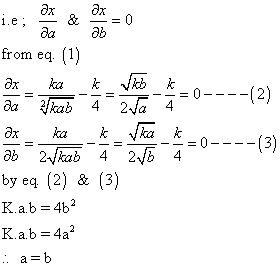
Note: this valid only when k<4 because if a = b Eq.(1)
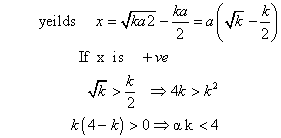
Equilibrium
Physical Eqn.
Low of mass action
Law of chemical Eqn.
Eqn.constant
Revessible rxn.
Forward rxn
Backward rxn
Gibbs free energy and Spontaneity
Homogeneous Equilibrium
Heterogeneous Equilibrium Le chatelier’s principleSource: goiit.com
- Dumb question
- why we have taken kp (0.015)2?
Ans: K
pfor division of HI = 0.015
HI
K
p
= 0.015
2HIH2+ I2 Kp= (0.015)2x = 0.2
Again since HI decomposes rxn proceeds in forward directioal (by Leehatetics Principal)
But a = (P + A) X 0.2
also kp
for final eqn (3)
= 0.8(P + A)2But Kp
= 137.5 X 137.5
0.8(P + A)2
= 137.5 X 137.5
P + A = 153.73
A = 16.22
a = (p + A) X 0.2 = 153.73 X 0.2 = 30.75
Total pressure at eqn =PNH3+PHI+PH2+PI
=(P+A)+(P+A-a)+A/2+A/2
=2p+2A
=2(p+A)= 2x153.73
=307.46 mmHg
Q.2: when 1 - Pentyne (a)is treated with 4n atc. KOH at 175 it slowely get converted into an eqn mix of 1.3 % 1 pentyne 95.2% 2 pentyne(b) of 1,2 pentadiene ©eqn. was maintained at 175c calculate
for following eqn.
from calculated value of g1 and g2 indicate order of (a) (b)© write a reasonable rxn mechanism in showing all intermediates reading to (A)(B)©
= 16.178 KJ
Stability order for A & B is B > A
Similerly for B
C
So, stability order for B and Cis B > C
Total stability order is B><>A
Q3. N2& 02
combine at a giventemp to produce no. at eqn the yield of no is x% by volume. If x =
where k is is eqn constant given rxn. at given temp and a and b volume % of n2 &o2 respectively in intial pure mixture what should be the intial composition of reacting mixture in order that max. yield of nois ensured? also repo max value of k at which x is maximum?
‘x’ is maximum only when condition of maximum are fulful
Note: this valid only when k<4 because if a = b Eq.(1)
Equilibrium
Physical Eqn.
Low of mass action
Law of chemical Eqn.
Eqn.constant
Revessible rxn.
Forward rxn
Backward rxn
Gibbs free energy and Spontaneity
Homogeneous Equilibrium
Heterogeneous Equilibrium Le chatelier’s principle
Source: goiit.com
- Dumb question
- why we have taken kp (0.015)2?
Ans: K
pfor division of HI = 0.015
HI
K
p
= 0.015
2HIH2+ I2 Kp= (0.015)2x = 0.2
Again since HI decomposes rxn proceeds in forward directioal (by Leehatetics Principal)
But a = (P + A) X 0.2
also kp
for final eqn (3)
= 0.8(P + A)2But Kp
= 137.5 X 137.5
0.8(P + A)2
= 137.5 X 137.5
P + A = 153.73
A = 16.22
a = (p + A) X 0.2 = 153.73 X 0.2 = 30.75
Total pressure at eqn =PNH3+PHI+PH2+PI
=(P+A)+(P+A-a)+A/2+A/2
=2p+2A
=2(p+A)= 2x153.73
=307.46 mmHg
Q.2: when 1 - Pentyne (a)is treated with 4n atc. KOH at 175 it slowely get converted into an eqn mix of 1.3 % 1 pentyne 95.2% 2 pentyne(b) of 1,2 pentadiene ©eqn. was maintained at 175c calculate
for following eqn.
from calculated value of g1 and g2 indicate order of (a) (b)© write a reasonable rxn mechanism in showing all intermediates reading to (A)(B)©
= 16.178 KJ
Stability order for A & B is B > A
Similerly for B
C
So, stability order for B and Cis B > C
Total stability order is B><>A
Q3. N2& 02
combine at a giventemp to produce no. at eqn the yield of no is x% by volume. If x =
where k is is eqn constant given rxn. at given temp and a and b volume % of n2 &o2 respectively in intial pure mixture what should be the intial composition of reacting mixture in order that max. yield of nois ensured? also repo max value of k at which x is maximum?
‘x’ is maximum only when condition of maximum are fulful
Note: this valid only when k<4 because if a = b Eq.(1)
Equilibrium
Physical Eqn.
Low of mass action
Law of chemical Eqn.
Eqn.constant
Revessible rxn.
Forward rxn
Backward rxn
Gibbs free energy and Spontaneity
Homogeneous Equilibrium
Heterogeneous Equilibrium Le chatelier’s principle
Source: goiit.com
br />
A = 16.22
a = (p + A) X 0.2 = 153.73 X 0.2 = 30.75
Total pressure at eqn =PNH3+PHI+PH2+PI
=(P+A)+(P+A-a)+A/2+A/2
=2p+2A
=2(p+A)= 2x153.73
=307.46 mmHg
Q.2: when 1 - Pentyne (a)is treated with 4n atc. KOH at 175 it slowely get converted into an eqn mix of 1.3 % 1 pentyne 95.2% 2 pentyne(b) of 1,2 pentadiene ©eqn. was maintained at 175c calculate
for following eqn.
from calculated value of g1 and g2 indicate order of (a) (b)© write a reasonable rxn mechanism in showing all intermediates reading to (A)(B)©
= 16.178 KJ
Stability order for A & B is B > A
Similerly for B
C
So, stability order for B and Cis B > C
Total stability order is B><>A
Q3. N2& 02
combine at a giventemp to produce no. at eqn the yield of no is x% by volume. If x =
where k is is eqn constant given rxn. at given temp and a and b volume % of n2 &o2 respectively in intial pure mixture what should be the intial composition of reacting mixture in order that max. yield of nois ensured? also repo max value of k at which x is maximum?
‘x’ is maximum only when condition of maximum are fulful
Note: this valid only when k<4 because if a = b Eq.(1)
Equilibrium
Physical Eqn.
Low of mass action
Law of chemical Eqn.
Eqn.constant
Revessible rxn.
Forward rxn
Backward rxn
Gibbs free energy and Spontaneity
Homogeneous Equilibrium
Heterogeneous Equilibrium Le chatelier’s principle
Source: goiit.com