Calorimetry And Thermal Expansion - 1
Calorimetry And Thermal Expansion
Introduction
Calorimetry can be considered to be the earliest known principal of Heat and Thermodynamics.This was the introduction of the widely used principal of Conservation of Energy to mankind.
Thermal Expansion too find a great importance in engineering application.
Its knowledge should also be imported for efficient working of various machines.
Calorimetry and Thermas Expansion:-
Calorimetry:-
Heat:-
It is a form of energy which determines the change in thermas state of a body.
Heat flows from a body which has a higher temp. to the body which has lower temp.
Specific Heat:- Quantity of heat in calories required to raise the temp. of 1 gm. of that substance by 1
 C.
C.thermal Equilibrium:- Two or move bodies an said to be in then mal equilibrium with each other if threr is no folow of heat from one body to other when they are brought in contact with each other.
Zenoth Law of Thermobodynamics and Temperature [Secondary Information]:-
If system A is in thermal equilibrium with system B and system B is in thermal equilibrium with system C, then system A and C are in equilibrium with each other.
Dum Question:-
Q. Why iron becomes hot in sunlight?
A. Heat flows from surroundings to iron. (high to lower temerrantance).
Temperature:- It is digree of hotness or coldness of body.
Measurement of Temprature [ Secondary Information]:- Temprature of a system is measured indivectly making use of the fact that cutain physical quantities of substance change with temperatire.
For eg, On celsius scale lo and l100 denote the length of the mercury at 0
 C and 100
C and 100 C resp.
C resp.We have
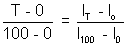
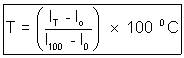
Similarly
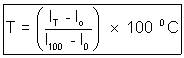
Where R0, RT, R100 denote electrical resistance at 0 0C, 100 0C, resp.
Triple Point[ Secondary Information]:- Unique set of pressure and temp. Exist at which are these states (solid, liquid and vapones) of a pure substance can exist orace in equilibrium. This temprature is called triple point of substance.
Kelvin Scale:
The celsius scale i9s defined such as
| T (K) = T (0C) + 273.15 |
Dum Question:-
Q. Some physical quantities change due to change in temp. Name a few.
A. Pressure, Volume, length, etc.
Q. The pressure of pure water is P at triple point. Temperature is increased to its boiling point. Pressure changes. What is the Changes.
A. Pressuer increases.
Illustration:
Q. When a block metal of specific heat 0.1 cal/g/ 0C and weighing 110 gm. is heated to 100 0C and then quickly transfered to a colarimeter containing 200 gm. if a liquid at 10 0C, the resulting temprature is 18 0C. On repeating the experiment with 400 gm. of same luquid in the same calorimeter at same initial temp., the resulting temp. is 14.5 0C. Find:
(a) Sp. heat of the liquid.
(b) The water equivalent of calorimeter.
Ans.
Heat lost by the block = Heat gained by (liquid + calorimeter)
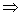 110 X 0.1 X (100 - 18) = 200 X S X (18 - 10) + W X (18 - 10)
110 X 0.1 X (100 - 18) = 200 X S X (18 - 10) + W X (18 - 10)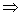 1600S + 8W = 902
1600S + 8W = 902Heat lost by the block = Heat gained by (liquid + Container)
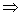 110 X 0.1 X (100 - 14.5) = 400 X S X (14.5 - 10) + W X (14.5 X10)
110 X 0.1 X (100 - 14.5) = 400 X S X (14.5 - 10) + W X (14.5 X10)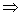 1800S + 14.5W = 940.5
1800S + 14.5W = 940.5S = 0.45 cal/g/C and W = 16.6 gm.
Thermal Expension:- When a solid is heated, its length, surface area and the volume increases.
Line as Expansion:
Consider a lod of length l1 at a temp. T1 - Let it be heated to a temp t2 and increased length of the rod be l2, then
l2 = l1(1 + 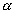 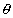 |
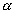 : Coefficient of linar expansion
: Coefficient of linar expansion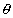 : T2 - T1
: T2 - T1[ Secondary Information]
We have dl
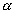 l>br /> and dl
l>br /> and dl 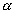 d
d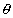
dl =
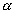 ld
ld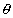
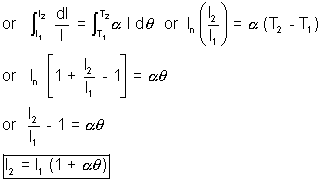
Superficial Expansion (Expansion of sunface):-
If A1 is the area of solid at T1 and A2 si the area at T2 , then
A2 = A1 (1 + 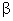 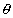 ) ) |
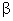 = Coefficient of superficial (Oval) Expansion
= Coefficient of superficial (Oval) Expansion