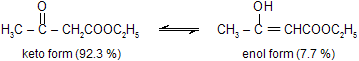Isomerism-1
Isomerism
The existence of two or more compounds with same molecular formula but different properties (physical, chemical or both) is known as isomerism; and the compounds themselves are called isomers. The term was given by Berzelius. The difference in properties of two isomers is due to the difference in the arrangement of atoms within their molecules. Isomerism may be of two types:
Structural isomerism
When the isomers differ only in the arrangement of atoms or groups within the molecule, without any reference to space, these are known as structural isomers and the phenomenon as structural isomerism. Thus the structural isomers have the same molecular formula, but possess different structural formulae. Structural isomerism may again be of several types.
(i) Chain, nuclear or skeleton isomerism
This type of isomerism is due to the difference in the nature of the carbon chain (i.e. straight or branched) which forms the nucleus of the molecule, e.g.,
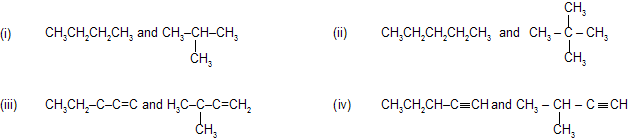
(ii) Position isomerism
It is due to the difference in the position of the substituent atom or group or an unsaturated linkage in the same carbon chain. Examples are
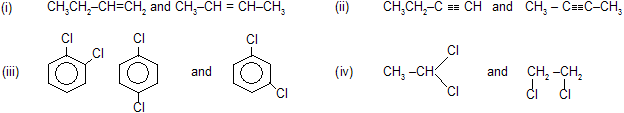
(iii) Functional isomerism
This type of isomerism is due to difference in the nature of functional group present in the isomers, e.g.,
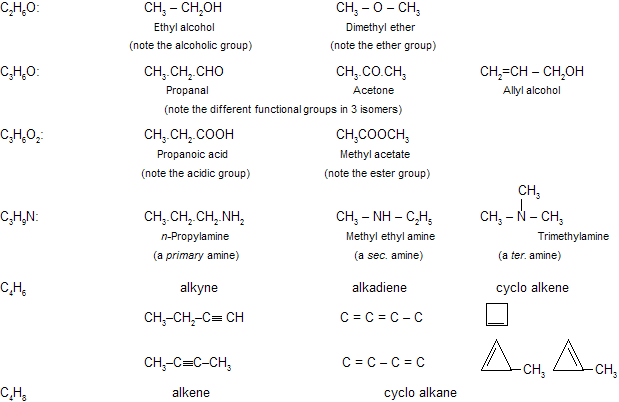
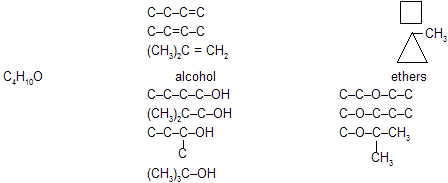 |
(iv) Metamerism
It is due to the difference in nature of alkyl groups attached to the same functional group. This type of isomerism is shown by compounds of the same homologous series. For example,
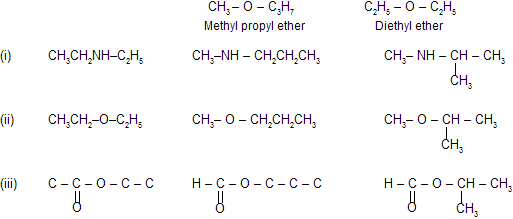
(v) Tautomerism
Tautomerism may be defined as the phenomenon in which a single compound exists in two readily interconvertible structures that differ markedly in the relative position of at least one atomic nucleus, generally hydrogen. The two different structures are known as tautomers of each other.
Sometimes the term tautomerism is also called as desmotropism (Greek desmos-bond; tropos-turn), since the interconversion of the two forms involves a change of bonds or dynamic isomerism as the two forms are in dynamic equilibrium with each other. Other names for tautomerism are kryptomerism, allelotropism or merotropy; however, tautomerism is the most widely accepted term.
There are several types of tautomerism of which keto-enol tautomerism is the most important. In this type, one form (tautomer) exists as a ketone while the other exists as an enol. The two simplest examples are of acetone and phenol.
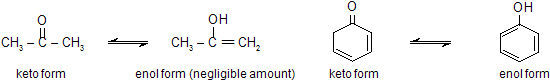
However, the most widely studied example of keto-enol tautomerism is that of acetoacetic ester (ethyl acetoacetate).